Last updated on February 5th, 2024 at 08:14 pm
URGENT RECALL: On Friday, October 14, 2022, the manufacturer Abbott initiated a proactive, voluntary recall of the following lots of 2 oz/59ml bottles of Ready-to-Feed liquid products for infants and children.
Which Similac and Other Abbott Products Are Included in the Recall
- Similac® Pro-Total ComfortTM
- Similac® 360 Total Care®
- Similac 360 Total Care Sensitive
- Similac® Special Care® 24, Similac Stage 1
- Similac® NeoSure®
- Similac Water (Sterilized)
- Pedialyte Electrolyte Solution
For a full list of LOT NUMBERS included in the Similac recall, please click here.

Why Were These Similac Products Recalled
These products are being recalled because a small percentage of products in the recalled lots have bottle caps that may not have sealed properly and completely, which could result in spoilage of the product. If spoiled product is consumed, gastrointestinal symptoms such as diarrhea and vomiting may occur.
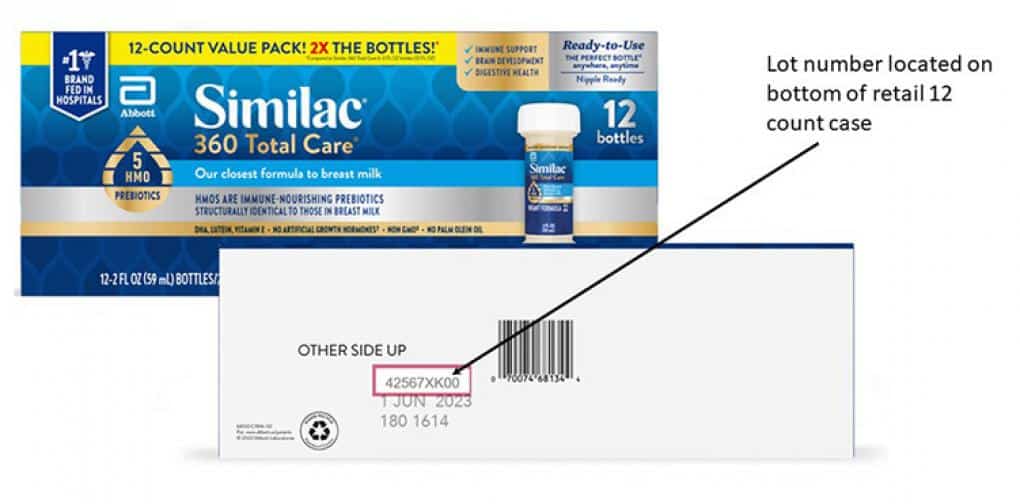
Check your products to see if your lot number was recalled. For a full list of recalled LOT NUMBERS, click here.
Where Is the Affected Area for the Recall
The products included in the Similac recall were manufactured at their Columbus, Ohio, manufacturing facility. The products included in the recall were distributed primarily to hospitals and to some doctors’ offices, distributors and retailers in the US, including Puerto Rico; one lot of products was sent to Barbados, Bermuda, Colombia, the Dominican Republic, Haiti, Jamaica, St. Croix and St. Thomas; and two lots were sent to Canada, Curacao, Panama, and Trinidad and Tobago.
Check Your Lot Numbers to See if Your Similac Was Recalled
To identify if your product is included, please visit similacrecall.com to view a list of impacted lot numbers or use the lot number checker on the webpage.
NOTE: The Similac recall is not expected to affect US Infant Formula Supply. This recall does not include any other liquid or powder formula brands or other nutrition products produced in the Columbus facility or elsewhere within their global nutrition manufacturing network. It also does not include any amino acid-based formulas or metabolic nutrition formulas.
What to Do if Your Product Is Included in the Recall
Do not use the product if you have a recalled version. For all feeding-related questions or questions about your child’s health, contact a healthcare professional.
If you have questions, please contact Abbott’s Consumer Relations hotlines below in the country where you received product:
- U.S.: +1-800-986-8540
- Puerto Rico: +1-787-622-5454
- Barbados: 246-417-0777
- Bermuda: 279-5568
- Canada: 1-855-733-4201
- Colombia: 1-800-518-9379
- Curacao: 737-2222 Ext. 235, 205
- Dominican Republic: +1-849-200-1564
- Haiti: 868-687-0223
- Jamaica: 876-927-7098
- Panama: 800-0410
- St. Croix: 1(340) 690-7222
- St. Thomas: 1(340) 690-7222
- Trinidad & Tobago: 868-687-0223
For more information about the recall, click here to visit the FDA’s page for this recall or go to www.similacrecall.com.










